Compound-Nucleus Reactions
When an incident particle approaches a target nucleus with impact parameter smaller than the nuclear radius, it can interact strongly with individual nucleons. After the initial encounter, the incident particle and recoiling nucleon undergo successive collisions inside the nucleus, progressively redistributing energy among many degrees of freedom. With small probability, a nucleon (or light cluster) acquires sufficient energy to escape, in close analogy with evaporation from a heated liquid.
Reaction Scheme and Two-Step Mechanism
The compound-nucleus picture represents the reaction as the formation of an intermediate, highly excited nucleus followed by its decay:
\[a + X \to C^* \to Y + b\]Here $C^*$ denotes the compound nucleus. A compact notation for the overall process is
\[X(a,b)Y\]The mechanism is conventionally separated into:
- Formation of the compound nucleus $C^*$.
- Decay of $C^*$ into a particular exit channel.
A central assumption is that the relative probability for decay into specific final products is independent of how $C^*$ was formed. Under this hypothesis, decay branching depends only on the total excitation energy and is governed by statistical considerations.
Illustrative Example and Cross-Section Systematics
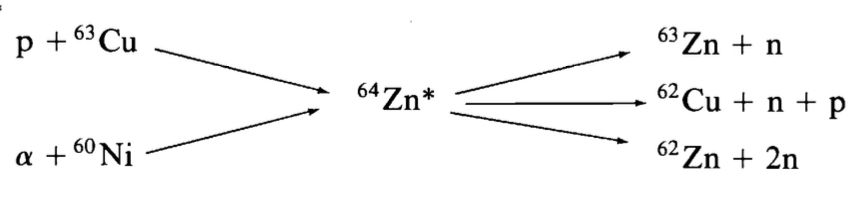
A representative case is the compound nucleus $^{64}\mathrm{Zn}^*$, which may be formed through distinct entrance channels such as
- $p + {}^{63}\mathrm{Cu}$,
- $\alpha + {}^{60}\mathrm{Ni}$.
Possible decay modes include, for example,
- ${}^{63}\mathrm{Zn} + n$,
- ${}^{62}\mathrm{Zn} + 2n$,
- ${}^{62}\mathrm{Cu} + p + n$.
If the compound-nucleus hypothesis holds, reactions that populate the same compound nucleus at the same excitation energy should exhibit similar relative cross-sections for corresponding decay channels. Thus, processes such as ${}^{63}\mathrm{Cu}(p,n){}^{63}\mathrm{Zn}$ and ${}^{60}\mathrm{Ni}(\alpha,n){}^{63}\mathrm{Zn}$ are expected to align when the incident energies are chosen to produce the same excitation energy in $^{64}\mathrm{Zn}^*$.
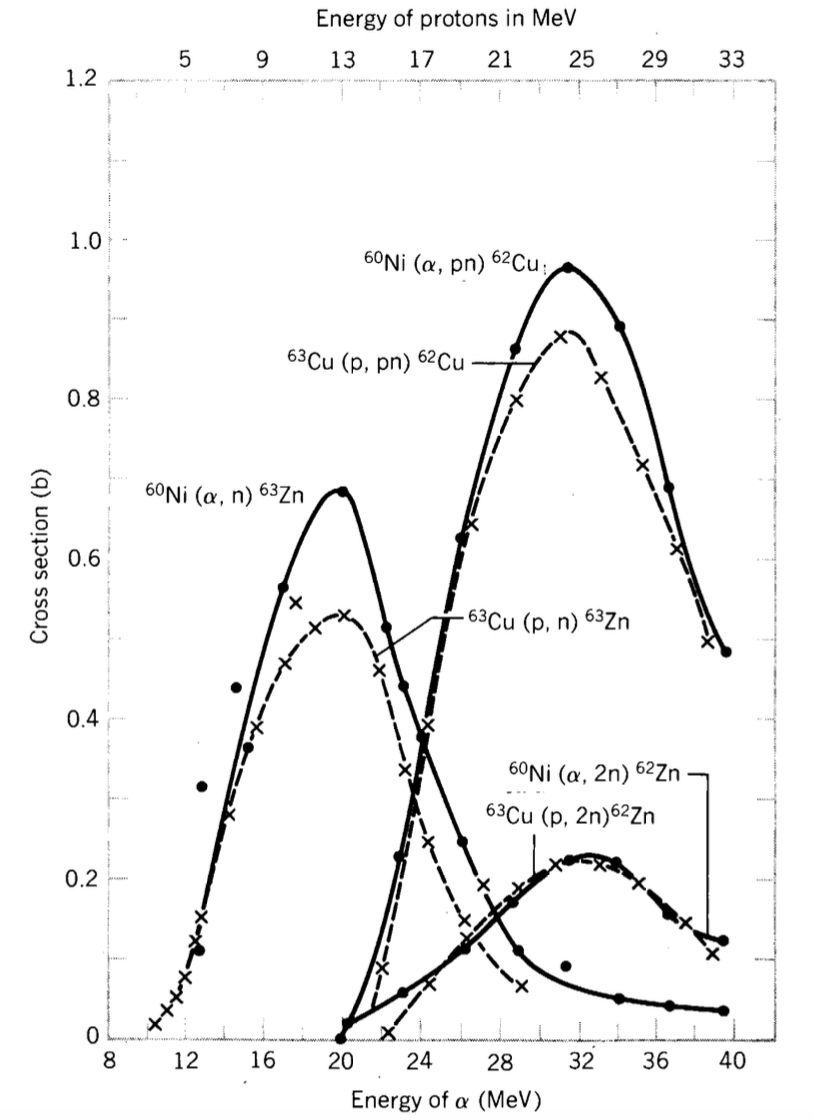
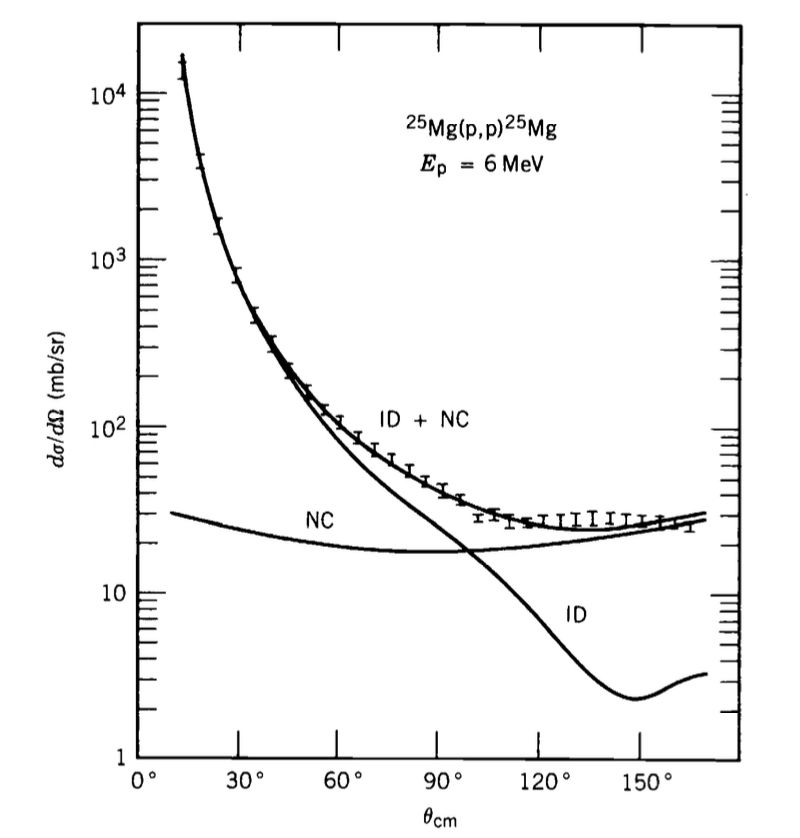
Conditions and Characteristic Angular Behaviour
The model is most effective when the incident energy is low enough that the projectile is unlikely to escape intact, typically in the range
\[10\text{--}20\ \mathrm{MeV}\]It is also most applicable to medium-weight and heavy nuclei, where the nuclear system can efficiently absorb and randomize the incident energy.
Because the decay proceeds after substantial equilibration, emission is generally close to isotropic in angle. A notable exception arises for heavy-ion reactions with significant angular momentum transfer, where preferential emission may appear near $0^\circ$ and $180^\circ$.
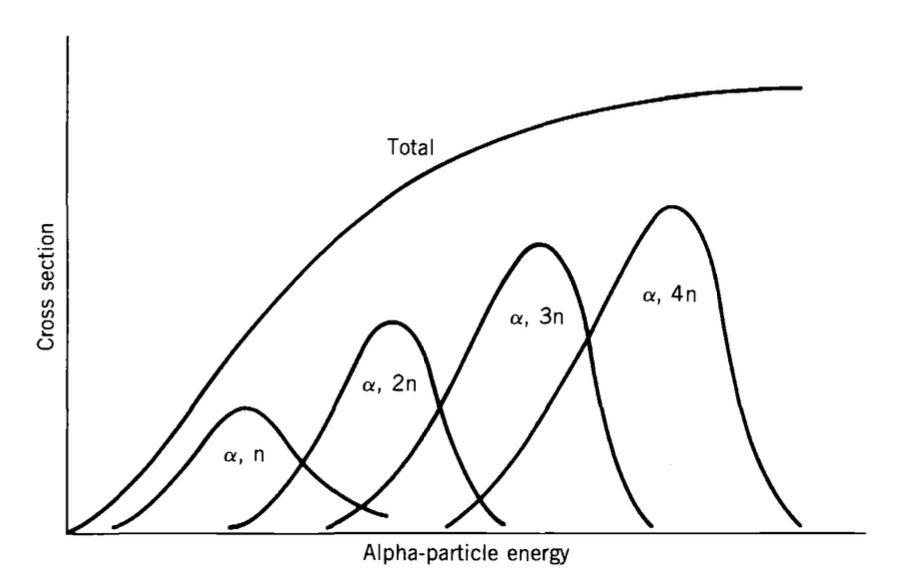
Energy Dependence and the Evaporation Analogy
In the evaporation picture, increasing excitation energy raises the likelihood of emitting additional particles. Consequently, channels of the form $(a,xn)$ typically show a rise of cross-section to a maximum, followed by a decline as still higher excitation energies favor emission of more particles (shifting strength into channels with larger multiplicity).